Smiley's Family Medicine Clinic - University of Minnesota Physicians, 2020 East 28th Street, Minneap
© Image by Gerd Altmann from Pixabay
According to a written report that examined how informed consent is given to COVID-xix vaccine trial participants, disclosure forms neglect to inform volunteers that the vaccine might make them susceptible to more severe disease if they're exposed to the virus.
The written report,1 "Informed Consent Disclosure to Vaccine Trial Subjects of Risk of COVID-19 Vaccine Worsening Clinical Disease," published in the International Periodical of Clinical Practice, Oct 28, 2020, points out that "COVID-19 vaccines designed to elicit neutralizing antibodies may sensitize vaccine recipients to more severe disease than if they were not vaccinated."
"Vaccines for SARS, MERS and RSV take never been approved, and the information generated in the evolution and testing of these vaccines suggest a serious mechanistic business organisation: that vaccines designed empirically using the traditional approach (consisting of the unmodified or minimally modified coronavirus viral fasten to elicit neutralizing antibodies), be they composed of protein, viral vector, DNA or RNA and irrespective of commitment method, may worsen COVID-19 disease via antibody-dependent enhancement (ADE)," the paper states.
"This adventure is sufficiently obscured in clinical trial protocols and consent forms for ongoing COVID-19 vaccine trials that adequate patient comprehension of this risk is unlikely to occur, obviating truly informed consent past subjects in these trials.
The specific and meaning COVID-xix risk of ADE should have been and should be prominently and independently disclosed to research subjects currently in vaccine trials, too equally those being recruited for the trials and future patients after vaccine blessing, in order to run into the medical ideals standard of patient comprehension for informed consent."
What Is Antibiotic-Dependent Enhancement?
Equally noted past the authors of that International Periodical of Clinical Exercise paper, previous coronavirus vaccine efforts — for severe acute respiratory syndrome coronavirus (SARS-CoV), Middle East respiratory syndrome coronavirus (MERS-CoV) and respiratory syncytial virus (RSV) — have revealed a serious concern: The vaccines have a tendency to trigger antibiotic-dependent enhancement.
What exactly does that mean? In a nutshell, it means that rather than raise your immunity confronting the infection, the vaccine actually enhances the virus' power to enter and infect your cells, resulting in more astringent affliction than had you not been vaccinated. 2
This is the verbal opposite of what a vaccine is supposed to do, and a meaning problem that has been pointed out from the very beginning of this push for a COVID-19 vaccine. The 2003 review paper "Antibody-Dependent Enhancement of Virus Infection and Disease" explains it this way:three
"In full general, virus-specific antibodies are considered antiviral and play an important role in the control of virus infections in a number of ways. However, in some instances, the presence of specific antibodies tin can exist beneficial to the virus. This activity is known as antibody-dependent enhancement (ADE) of virus infection.
The ADE of virus infection is a miracle in which virus-specific antibodies heighten the entry of virus, and in some cases the replication of virus, into monocytes/macrophages and granulocytic cells through interaction with Fc and/or complement receptors.
This miracle has been reported in vitro and in vivo for viruses representing numerous families and genera of public health and veterinary importance. These viruses share some common features such every bit preferential replication in macrophages, ability to found persistence, and antigenic variety. For some viruses, ADE of infection has go a swell business to disease command past vaccination."
Previous Coronavirus Vaccine Efforts Have All Failed
In my May 2020 interview above with Robert Kennedy Jr., he summarized the history of coronavirus vaccine evolution, which began in 2002, following three consecutive SARS outbreaks. By 2012, Chinese, American and European scientists were working on SARS vaccine development, and had about 30 promising candidates.
Of those, the iv all-time vaccine candidates were then given to ferrets, which are the closest analogue to human lung infections. In the video beneath, which is a select outtake from my full interview, Kennedy explains what happened next. While the ferrets displayed robust antibiotic response, which is the metric used for vaccine licensing, in one case they were challenged with the wild virus, they all became severely ill and died.
The aforementioned thing happened when they tried to develop an RSV vaccine in the 1960s. RSV is an upper respiratory illness that is very like to that caused by coronaviruses. At that time, they had decided to skip animate being trials and get direct to human being trials.
"They tested it on I think almost 35 children, and the same matter happened," Kennedy said. "The children developed a champion antibiotic response — robust, durable. It looked perfect [simply when] the children were exposed to the wild virus, they all became ill. Two of them died. They abandoned the vaccine. It was a large embarrassment to FDA and NIH."
Neutralizing Versus Bounden Antibodies
Coronaviruses produce not but ane but two different types of antibodies:
- Neutralizing antibodies,4 besides referred to equally immunoglobulin G (IgG) antibodies, that fight the infection
- Binding antibodiesfive (also known every bit non-neutralizing antibodies) that cannot prevent viral infection
Instead of preventing viral infection, binding antibodies trigger an aberrant immune response known as "paradoxical immune enhancement." Some other way to look at this is your immune arrangement is actually backfiring and non functioning to protect you but actually making you worse.
Many of the COVID-19 vaccines currently in the running are using mRNA to instruct your cells to make the SARS-CoV-2 spike poly peptide (S poly peptide). The spike protein, which is what attaches to the ACE2 receptor of the cell, is the beginning phase of the two-phase process viruses apply to gain entry into cells.
The idea is that by creating the SARS-CoV-2 spike protein, your immune system will commence production of antibodies, without making you lot sick in the process. The key question is, which of the 2 types of antibodies are being produced through this process?
Without Neutralizing Antibodies, Expect More Severe Illness
In an Apr 2020 Twitter thread,6 The Immunologist noted: "While developing vaccines ... and because amnesty passports, we must kickoff understand the complex part of antibodies in SARS, MERS and COVID-nineteen." He goes on to list several coronavirus vaccine studies that have raised concerns virtually ADE.
The start is a 2017 study7 in PLOS Pathogens, "Enhanced Inflammation in New Zealand White Rabbits When MERS-CoV Reinfection Occurs in the Absence of Neutralizing Antibody," which investigated whether getting infected with MERS would protect the bailiwick against reinfection, every bit is typically the case with many viral illnesses. (Meaning, once you recover from a viral infection, say measles, y'all're allowed and won't contract the illness again.)
To determine how MERS affects the immune system, the researchers infected white rabbits with the virus. The rabbits got sick and developed antibodies, but those antibodies were non the neutralizing kind, meaning the kind of antibodies that cake infection. As a effect, they were not protected from reinfection, and when exposed to MERS for a second time, they became ill again, and more severely and so.
"In fact, reinfection resulted in enhanced pulmonary inflammation, without an associated increment in viral RNA titers," the authors noted. Interestingly, neutralizing antibodies were elicited during this second infection, preventing the animals from being infected a 3rd fourth dimension. Co-ordinate to the authors:
"Our data from the rabbit model suggests that people exposed to MERS-CoV who fail to develop a neutralizing antibody response, or persons whose neutralizing antibody titers have waned, may be at gamble for severe lung disease on re-exposure to MERS-CoV."
In other words, if the vaccine does not upshot in a robust response in neutralizing antibodies, you might exist at gamble for more than severe lung disease if you're infected with the virus.
And here's an important signal: COVID-19 vaccines are Not designed to prevent infection. As detailed in "How COVID-xix Vaccine Trials Are Rigged," a "successful" vaccine but needs to reduce the severity of the symptoms. They're not fifty-fifty looking at reducing infection, hospitalization or death rates.
ADE in Dengue Infections
The Dengue virus is also known to cause ADE. As explained in a Swiss Medical Weekly paper published in April 2020:8
"The pathogenesis of COVID-19 is currently believed to go along via both directly cytotoxic and immune-mediated mechanisms. An additional mechanism facilitating viral cell entry and subsequent damage may involve the so-chosen antibody-dependent enhancement (ADE).
ADE is a very well-known cascade of events whereby viruses may infect susceptible cells via interaction between virions complexed with antibodies or complement components and, respectively, Fc or complement receptors, leading to the amplification of their replication.
This phenomenon is of enormous relevance not only for the agreement of viral pathogenesis, only likewise for developing antiviral strategies, notably vaccines ...
There are four serotypes of Dengue virus, all eliciting protective immunity. Notwithstanding, although homotypic protection is long-lasting, cantankerous-neutralizing antibodies against different serotypes are short-lived and may final only up to 2 years.
In Dengue fever, reinfection with a different serotype runs a more severe form when the protective antibiotic titer wanes. Here, not-neutralizing antibodies have over neutralizing ones, bind to Dengue virions, and these complexes mediate the infection of phagocytic cells via interaction with the Fc receptor, in a typical ADE.
In other words, heterotypic antibodies at subneutralizing titres account for ADE in persons infected with a serotype of Dengue virus that is different from the first infection.
Cross-reactive neutralizing antibodies are associated with decreased odds of symptomatic secondary infection, and the higher the titer of such antibodies following the primary infection, the longer the delay to symptomatic secondary infection ..."
The newspaper goes on to detail results from follow-up investigations into the Dengue vaccine, which revealed the hospitalization rate for Dengue among vaccinated children under the historic period of nine was greater than the rate among controls. The explanation for this appears to exist that the vaccine mimicked a primary infection, and equally that immunity waned, the children became susceptible to ADE when they encountered the virus a second time. The author explains:
"A post hoc analysis of efficacy trials, using an anti-nonstructural protein 1 immunoglobulin G (IgG) enzyme-linked immunosorbent analysis (ELISA) to distinguish antibodies elicited past wild-type infection from those following vaccination, showed that the vaccine was able to protect against severe Dengue [in] those who had been exposed to the natural infection before vaccination, and that the hazard of severe clinical outcome was increased among seronegative persons.
Based on this, a Strategic Counselor Group of Experts convened past World Health Organization (WHO) concluded that only Dengue seropositive persons should be vaccinated whenever Dengue control programs are planned that include vaccination."
ADE in Coronavirus Infections
This could finish up beingness of import for the COVID-nineteen vaccine. Hypothetically speaking, if SARS-CoV-2 works similar Dengue, which is as well caused by an RNA virus, and so anyone who has non tested positive for SARS-CoV-ii might actually be at increased chance for severe COVID-nineteen later on vaccination, and only those who take already recovered from a tour of COVID-nineteen would be protected against severe disease by the vaccine.
To be clear, we do not know whether that is the example or non, but these are of import areas of inquiry and the current vaccine trials will simply not be able to answer this of import question.
The Swiss Medical Weekly paper 9 besides reviews the evidence of ADE in coronavirus infections, citing inquiry showing inoculating cats against the feline infectious peritonitis virus (FIPV) — a feline coronavirus — increases the severity of the illness when challenged with the same FIPV serotype as that in the vaccine.
Experiments accept shown immunization with a variety of SARS vaccines resulted in pulmonary immunopathology one time challenged with the SARS virus.
The paper also cites inquiry showing "Antibodies elicited past a SARS-CoV vaccine enhanced infection of B prison cell lines in spite of protective responses in the hamster model." Another paper,10 "Antibody-Dependent SARS Coronavirus Infection Is Mediated by Antibodies Against Spike Proteins," published in 2014, establish that:
"... higher concentrations of anti-sera confronting SARS-CoV neutralized SARS-CoV infection, while highly diluted anti-sera significantly increased SARS-CoV infection and induced higher levels of apoptosis.Results from infectivity assays indicate that SARS-CoV ADE is primarily mediated by diluted antibodies against envelope fasten proteins rather than nucleocapsid proteins. We also generated monoclonal antibodies confronting SARS-CoV fasten proteins and observed that most of them promoted SARS-CoV infection.
Combined, our results suggest that antibodies against SARS-CoV spike proteins may trigger ADE effects. The data raise new questions regarding a potential SARS-CoV vaccine ..."
A study11 that ties into this was published in the periodical JCI Insight in 2019. Here, macaques vaccinated with a modified vaccinia Ankara (MVA) virus encoding full-length SARS-CoV spike protein ended up with more than severe lung pathology when the animals were exposed to the SARS virus. And, when they transferred anti-spike IgG antibodies into unvaccinated macaques, they developed acute diffuse alveolar damage, likely by "skewing the inflammation-resolving response."
SARS Vaccine Worsens Infection After Challenge With SARS-CoV
An interesting 2012 newspaper 12 with the telling title, "Immunization with SARS Coronavirus Vaccines Leads to Pulmonary Immunopathology on Challenge with the SARS Virus," demonstrates what many researchers now fear, namely that COVID-19 vaccines may end upwards making people more prone to severe SARS-CoV-ii infection.
The newspaper reviews experiments showing immunization with a multifariousness of SARS vaccines resulted in pulmonary immunopathology once challenged with the SARS virus. As noted past the authors: 13
Inactivated whole virus vaccines whether inactivated with formalin or beta propiolactone and whether given with our without alum adjuvant exhibited a Th2-type immunopathologic in lungs after challenge.As indicated, two reports attributed the immunopathology to presence of the Due north protein in the vaccine; notwithstanding, nosotros establish the same immunopathologic reaction in animals given S protein vaccine only, although it appeared to be of lesser intensity.
Thus, a Th2-type immunopathologic reaction on challenge of vaccinated animals has occurred in three of 4 animal models (not in hamsters) including two different inbred mouse strains with four dissimilar types of SARS-CoV vaccines with and without alum adjuvant. An inactivated vaccine preparation that does non induce this event in mice, ferrets and nonhuman primates has not been reported.
This combined experience provides business organization for trials with SARS-CoV vaccines in humans. Clinical trials with SARS coronavirus vaccines have been conducted and reported to induce antibiotic responses and to be 'safe.' Still, the evidence for safety is for a short menstruum of observation.
The concern arising from the present written report is for an immunopathologic reaction occurring among vaccinated individuals on exposure to infectious SARS-CoV, the footing for developing a vaccine for SARS. Boosted condom concerns relate to effectiveness and safety confronting antigenic variants of SARS-CoV and for safety of vaccinated persons exposed to other coronaviruses, particularly those of the type 2 group."
The Elderly Are Most Vulnerable to ADE
On pinnacle of all of these concerns, in that location's show showing the elderly — who are most vulnerable to astringent COVID-nineteen — are as well the most vulnerable to ADE. Preliminary research findings14 posted on the preprint server medRxiv at the end of March 2020 reported that eye-aged and elderly COVID-19 patients take far higher levels of anti-spike antibodies — which, again, increase infectivity — than younger patients.
Immune Enhancement Is a Serious Concern
Another paper worth mentioning is the May 2020 mini reviewfifteen "Impact of Immune Enhancement on COVID-19 Polyclonal Hyperimmune Globulin Therapy and Vaccine Development." As in many other papers, the authors point out that:16
"While development of both hyperimmune globulin therapy and vaccine against SARS-CoV-ii are promising, they both pose a mutual theoretical safe business concern. Experimental studies have suggested the possibility of immune-enhanced affliction of SARS-CoV and MERS-CoV infections, which may thus similarly occur with SARS-CoV-ii infection ...
Immune enhancement of affliction can theoretically occur in ii means. Firstly, non-neutralizing or sub-neutralizing levels of antibodies can enhance SARS-CoV-two infection into target cells.
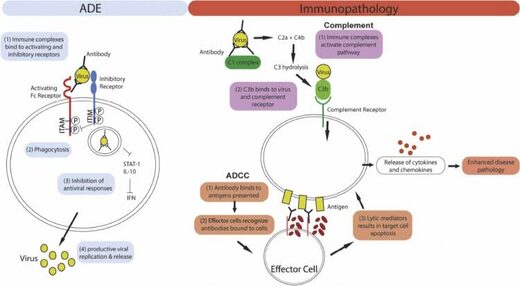
Secondly, antibodies could enhance inflammation and hence severity of pulmonary illness. An overview of these antibody dependent infection and immunopathology enhancement effects are summarized in Fig. i ...
Currently, in that location are multiple SARS-CoV and MERS-CoV vaccine candidates in pre-clinical or early on phase clinical trials. Animal studies on these CoVs have shown that the spike (South) protein-based vaccines (specifically the receptor binding domain, RBD) are highly immunogenic and protective confronting wild-type CoV challenge.
Vaccines that target other parts of the virus, such as the nucleocapsid, without the Southward poly peptide, have shown no protection against CoV infection and increased lung pathology. However, immunization with some S poly peptide based CoV vaccines accept also displayed signs of enhanced lung pathology following challenge.
Hence, besides the choice of antigen target, vaccine efficacy and risk of immunopathology may be dependent on other ancillary factors, including adjuvant formulation, age at vaccination ... and route of immunization."
© manufactures.mercola.com
Figure ane: Mechanism of ADE and antibody mediated immunopathology. Left panel: For ADE, immune complex internalization is mediated by the engagement of activating Fc receptors on the cell surface. Co-ligation of inhibitory receptors then results in the inhibition of antiviral responses which leads to increased viral replication. Right panel: Antibodies tin can cause immunopathology past activating the complement pathway or antibody-dependent cellular cytotoxicity (ADCC). For both pathways, excessive immune activation results in the release of cytokines and chemokines, leading to enhanced illness pathology.
Do a Gamble-Benefit Analysis Before Making Up Your Listen
In all likelihood, regardless of how constructive (or ineffective) the COVID-19 vaccines terminate up being, they'll be released to the public in relatively short order. Most predict one or more than vaccines will be ready erstwhile in 2021.
Ironically, the data 17,18,xix we at present have no longer back up a mass vaccination mandate, considering the lethality of COVID-nineteen is lower than the flu for those under the age of sixty. 20 If you're under the age of 40, your risk of dying from COVID-xix is merely 0.01%, meaning you have a 99.99% chance of surviving the infection. And yous could amend that to 99.999% if y'all're metabolically flexible and vitamin D replete.
Then, really, what are nosotros protecting against with a COVID-19 vaccine? As mentioned, the vaccines aren't fifty-fifty designed to prevent infection, only reduce the severity of symptoms. Meanwhile, they could potentially make you sicker once you're exposed to the virus. That seems similar a lot of risk for a truly questionable benefit.
To circle back to where we started, participants in current COVID-19 vaccine trials are not being told of this adventure — that by getting the vaccine they may stop up with more astringent COVID-19 in one case they're infected with the virus.
Lethal Th2 Immunopathology Is Another Potential Hazard
In closing, consider what this PNAS news characteristic states about the gamble of vaccine-induced allowed enhancement and dysfunction, particularly for the elderly, the very people who would need the protection a vaccine might offer the most:21
"Since the 1960s, tests of vaccine candidates for diseases such as dengue, respiratory syncytial virus (RSV), and severe acute respiratory syndrome (SARS) accept shown a paradoxical phenomenon:
Some animals or people who received the vaccine and were subsequently exposed to the virus developed more severe affliction than those who had not been vaccinated. The vaccine-primed immune arrangement, in certain cases, seemed to launch a shoddy response to the natural infection ...
This immune backfiring, or and then-chosen immune enhancement, may manifest in dissimilar ways such as antibody-dependent enhancement (ADE), a process in which a virus leverages antibodies to aid infection; or cell-based enhancement, a category that includes allergic inflammation caused by Th2 immunopathology. In some cases, the enhancement processes might overlap ...
Some researchers argue that although ADE has received the well-nigh attending to date, it is less likely than the other immune enhancement pathways to crusade a dysregulated response to COVID-19, given what is known about the epidemiology of the virus and its behavior in the human being body.
'There is the potential for ADE, but the bigger problem is probably Th2 immunopathology,' says Ralph Baric, an epidemiologist and expert in coronaviruses ... at the Academy of North Carolina at Chapel Hill.
In previous studies of SARS, aged mice were found to have particularly loftier risks of life-threatening Th2 immunopathology ... in which a faulty T cell response triggers allergic inflammation, and poorly functional antibodies that class immune complexes, activating the complement system and potentially dissentious the airways."
Sources and References
- 1 International Journal of Clinical Practice, October 28, 2020 DOI: ten.111/ijcp.13795
- 2, 21 PNAS.org Apr 14, 2020 117 (xv) 8218-8221
- 3 Viral Immunology 2003;xvi(i):69-86
- four Science Direct Neutralizing Antibiotic
- 5 Science Direct Binding Antibody
- 6 Twitter, The Immunologist April 9, 2020
- 7 PLOS Pathogens 2017 Aug; 13(8): e1006565
- viii, 9 Swiss Medical Weekly April 16, 2020; 150:w20249
- 10 Biochemical and Biophysical Enquiry Communications August 22, 2014; 451(two): 208-214
- 11 JCI Insight February 21, 2019 DOI: ten.1172/jci.insight.123158
- 12 PLOS ONE April 2012; 7(four): e35421 (PDF)
- 13 PLOS I April 2012; 7(4): e35421 (PDF), page 11
- 14 medRxiv DOI:10.1101/2020.03.30.20047365 (PDF)
- 15 EBioMedicine 2020 May; 55: 102768
- sixteen EBioMedicine 2020 May; 55: 102768, Introduction
- 17, xx Annals of Internal Medicine September 2, 2020 DOI: ten.7326/M20-5352
- 18 YouTube, SARS-CoV-2 and the rising of medical technocracy, Lee Merritt, Medico, aprox viii minutes in (Prevarication No. 1: Expiry Risk)
- 19 Technical Report June 2020 DOI: 10.13140/RG.2.24350.77125
krogerreplads1943.blogspot.com
Source: https://www.sott.net/article/445095-How-COVID-19-vaccine-can-destroy-your-immune-system
0 Response to "Smiley's Family Medicine Clinic - University of Minnesota Physicians, 2020 East 28th Street, Minneap"
Post a Comment